INTRODUCING THE ALL NEW…

For Atraumatic Mesh Fixation in Open Inguinal Hernia Surgery

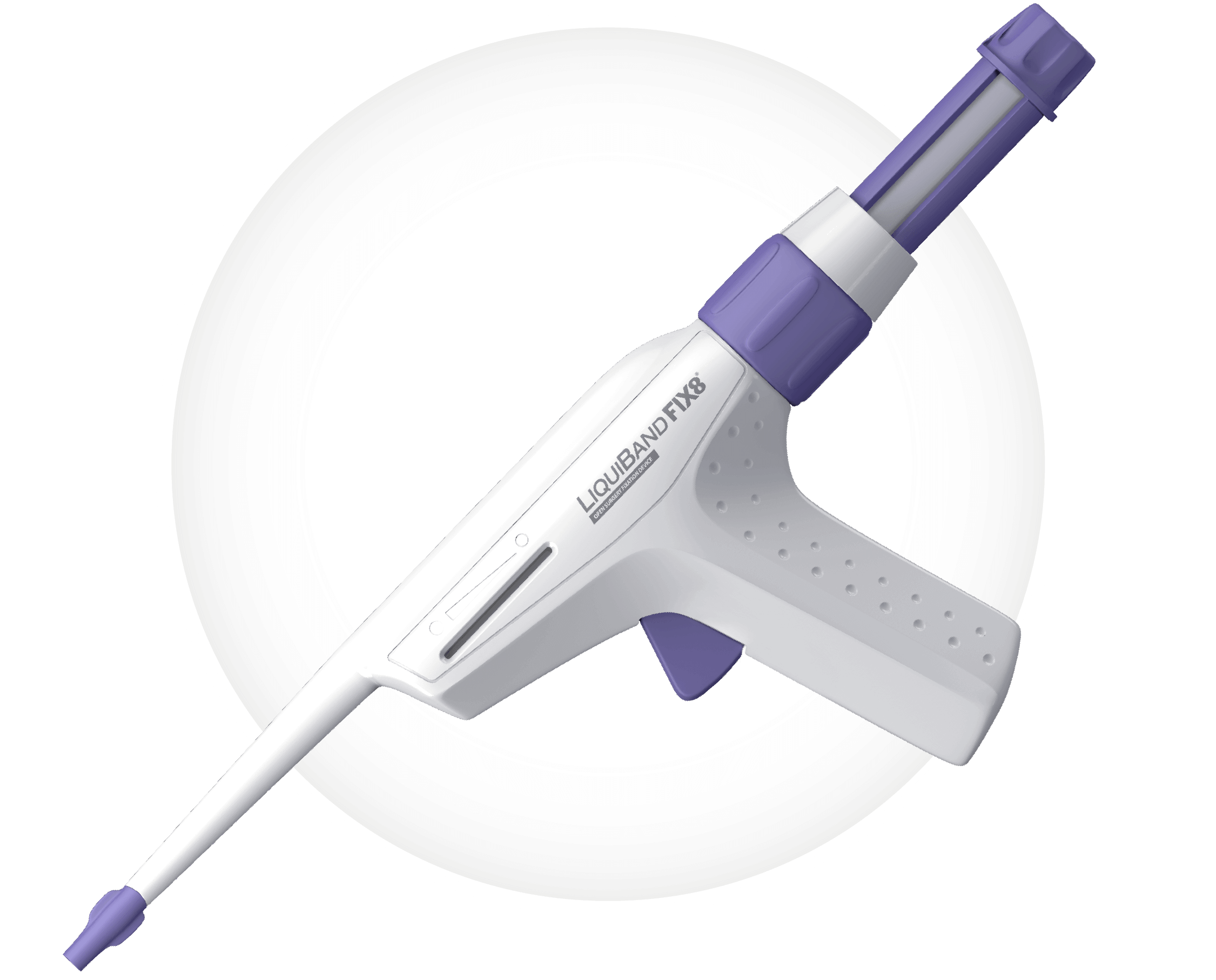
Key Features
1 Removable Cannula Tip
2 Volume indicator
3 Trigger
4 Priming Dial
5 Ampoule Plunger
6 Visualisation Window
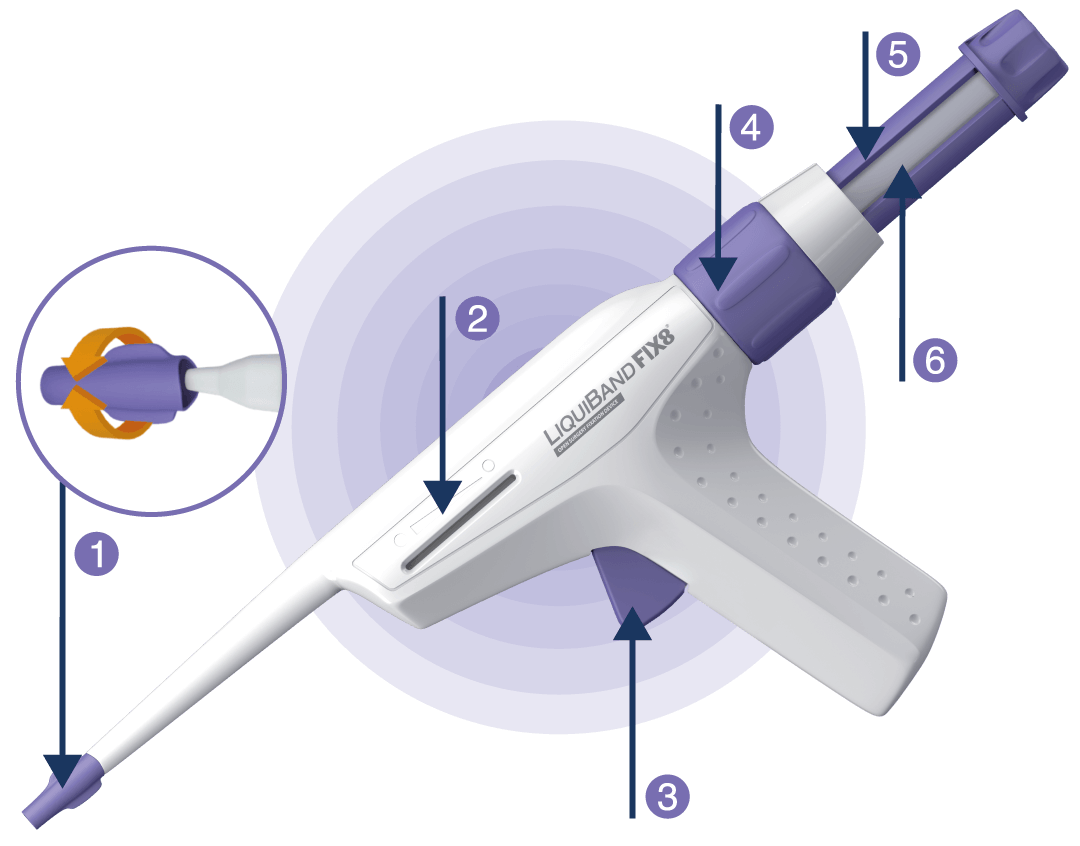
1 Removable Cannula Tip
2 Volume indicator
3 Trigger
4 Priming Dial
5 Priming Ampoule Plunger
6 Priming Visualisation Window
A unique dual tip atraumatic device engineered for strong mesh fixation & topical wound closure in open inguinal hernia surgery
Key Benefits
Innovative tip design 1
- Removable tip for topical wound closure and mesh fixation
- Designed to project liquid anchors drop by drop
- Guarded aperture to prevent tip blockage
Simplified Priming 2
- Simple priming process
- Pre assembled
- Easy tip removal for topical wound closure
Patient comfort
- N-Butyl 2 cyanoacrylate adhesive* can be considered as a good replacement for sutures in inguinal hernia repair leading to lesser post operative morbidities and a better quality of life. 8
N-Butyl 2 Cyanoacrylate adhesive
- Mesh fixation in 10 sec 3
- Reduced risk of trauma & post-operative complications 4
- Low exothermic reaction 5
Precise & Ergonomic 6
- Easy handling
- Delivers at least 45 liquid anchors
- Each trigger dispenses 12.5mg of adhesive
Cost Effectiveness
- N-Butyl 2 cyanoacrylate adhesive* optimises cost in open hernia procedure due to less turnaround time which results in improved theatre output without compromising patient care.
Performance
Strong Fixation
LiquiBand FIX8® Open demonstrated higher burst strength in comparison to mechanical mesh fixation devices.
N=6 in all groups (In vivo)
LiquiBand Fix8® Open Burst Strength vs Tacking Devices 7
Time Saving
Mesh fixation with N-Butyl 2 cyanoacrylate* is superior to sutures as glue reduces the operative time by an average of 10.8 minutes.8
| Group | Mean | SD | Mean±SD |
|---|---|---|---|
| Glue | 41.8 | 5.65 | 41.8±5.65 |
| Suture | 52.6 | 4.64 | 52.6±4.64 |
| Total | 47.29 | 7.46 | 47.29±7.46 |
Product Videos
Product Videos
LiquiBand FIX8® Open
Hernia Fixation Device Demonstration
Sizes & Codes
LiquiBand FIX8® Open is available through a variety of channels.
| Product | Product Code | NHS SC Code | Volume per Each | Each per Box |
|---|---|---|---|---|
| LiquiBand FIX8® OPEN | 72014021 | - | 1.5g | 6 |
